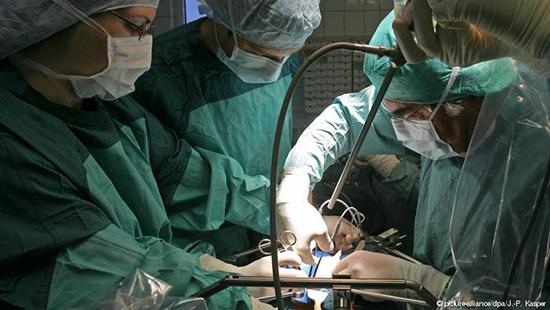
Surgeon Bruno Reichart has successfully implanted a pig’s heart into a baboon. He tells DW why harvesting from other species could address a chronic shortage of human donor organs.
Deutsche Welle: Mr. Reichart, transplanting a pig’s heart into a baboon for the first time offers hope this might be possible for humans, too. Why are pigs particularly suitable as donor animals?
Bruno Reichart: Ethics play an important role here. We’ve been eating pigs for a long time, so it’s socially acceptable to kill them. Also, pigs produce many offspring in a very short time — every four months — and they are fully grown and sexually mature after six months.
A pig’s heart is also very similar in structure to a human heart. The valves from pigs’ hearts have been used as a replacement in humans for 40 years.
And why baboons as recipients?
This is part of what the authorities demand: The organ should not be implanted into a pig or a dog but a primate that’s very close to us biologically, so conclusions can be drawn as to whether the intervention could also be successful in humans.
Could you use any ordinary pig as a donor?
You have to adapt the pig’s heart to that of a human in order to prevent the recipient’s body rejecting the transplant. Which is why the pig hearts are genetically modified before they are transplanted.
Just a few months ago critics argued there were too many to such transplants — saying that pig hearts lacked the pumping capacity of a human heart, and that porcine endogenous retroviruses (PERV) in the pig genome could put human patients at risk.
I have to say, the people who wrote this unfortunately had little idea what they were talking about and should have done a bit more reading. A pig’s heart pumps excellently in the body of a baboon or human.
There has never been an infection from viruses in pig tissue. There are three types of porcine endogenous retroviruses: A, B and C. C is very aggressive so we have to use animals that are C-negative, or don’t have C viruses due to breeding or genetic modification.
How does this genetic modification work?
This happens in the egg cell. You have to remove a gene, which is relatively easy nowadays with CRISPR-Cas9 scissors. You can use it to destroy the C-PERV copies in the pig genome and make them harmless.
What would be the advantage of a pig’s heart over current transplant options?
This would have the advantage of addressing the enormous shortage donors. And that’s our goal — that a pig’s heart isn’t just a bridging measure, but a final transplant.
Is this the moment of breakthrough?
There will have to be some more breakthroughs, I’m afraid. Now we need money, because these trials cost a lot. We have to find an investor, and that’s very difficult in Europe. For me, it’s currently a full-time job.
The Deutsche Forschungsgemeinschaft (German Research Foundation) is financing the trials very generously at the moment, but in order to carry out a pilot study we need additional financial resources and a network that also includes hospitals.
What would it mean if pig hearts became a real alternative? Would production farms suddenly appear everywhere?
At the moment, a few pigs and their offspring would be sufficient for the pilot study. And later, you would certainly need 1,000 pigs — this is still a long way off. The pigs already exist, but for organ transplants you would need very high hygiene standards. We don’t have such capabilities anywhere yet.
How can you be sure this will work?
You always have throw yourself into the unknown. But it’s very unlikely this will not work.
Bruno Reichart is professor emeritus at the University Hospital in Munich and one of the most distinguished heart transplant experts in Germany. In 1983, he successfully carried out the first heart-lung transplant in Germany. Today, he works on xenotransplantation — the transfer of cells, up to entire organs, between different species.
The interview was conducted by Anne Höhn.